R&D
- Mar 1, 2016
Galvanically Active Wires for Enhanced Absorbable Device Performance
J.E. Schaffer, Ph.D., Senior Research and Development Engineer & A.J. Griebel, Research and Development Engineer
Medical device design with absorbable metals has the potential to revolutionize patient care by providing effective short-term therapy and then harmlessly dissolving away. One of the primary hurdles to overcome is that of premature material fracture which could potentially lead to improper device function. One potential avenue to solve this problem is to harness and use a natural property of metals to our advantage, namely galvanic activity. When two differing metals are in proximity to one another in a conductive solution, one material will be electrochemically dissolved and the other protected from dissolving. This is the same principle by which a potato battery or galvanized steel works.
Galvanic activity in metals can be used to design safer absorbable medical devices that require high strength drawn wire by using wire composite technology to effectively control the degradation sequence. Absorbable alloy wires with strength levels approaching 2 GPa (>280,000 psi) have recently been engineered for prototype medical device service [1]. Even more intriguing is the fact that these strength levels were achieved in nutrient alloys containing elements commonly found on a cereal box nutrition label, making them well tolerated in human cell environments [2]. High strength and elasticity in drawn magnesium, iron and even zinc alloys and composites [3-5] are being designed into next generation devices that will provide therapy before harmlessly dissolving away.
The hand-off of mechanical load from a tissue-supporting implant to the body’s tissue is a key advantage of absorbable technology [6-7]. Hard and soft tissues operate best when mechanically unhindered and free to sense their environment. As a parallel, consider a cast for a broken bone: necessary and beneficial in the short term while the bone heals, but cumbersome and even harmful in the long run. Likewise, one important key to the success of absorbable endovascular, orthopedic, and other devices, is to ensure that their mechanical integrity is not handed-off to the body prematurely. Unique material constructs that harness electrochemical activity to promote beneficial device-tissue hand-off are under development by our team, several examples of which are illustrated in Figures 1-3 below. For more information, please contact: [email protected]. [email protected].
.png)
Figure 1 - Panel D shows the cross section of a 200 ?m magnesium alloy (LAEX6410) wire with a 65 ?m high purity (99.95%) iron core; this wire was processed to a strength level exceeding 600 MPa. Panel E shows a section of wire coil (F) that was exposed to body temperature serum.
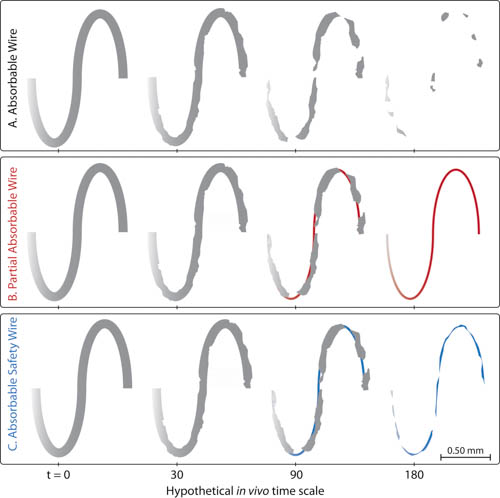
Figure 2 – Absorbable filament time series based on galvanically active design and experimental observation. (A) Fully absorbable filament may give rise to some debris over the course of device hand-off to tissue. Generation of these debris may not compromise device function if tissue has adequately enveloped the material. (B) A partially absorbable design with a more cathodic, non-degrading core can hand-off most of the mechanical load due to high loss of flexural rigidity, while reducing the risk of premature debris formation. (C) An absorbable safety element, that is somewhat more cathodic than the bulk structure, can provide both full absorption and reduced risk of premature debris formation.
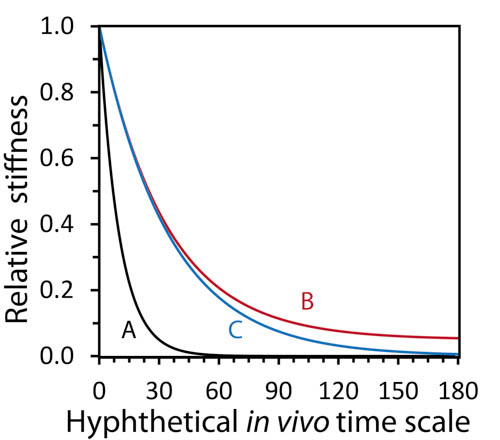
Figure 3 – Relative stiffness reduction in each case above over a hypothetical implant course.
Further Reading
[5] Bowen, Patrick K., Jaroslaw Drelich, and Jeremy Goldman. "Zinc exhibits ideal physiological corrosion behavior for bioabsorbable stents." Adv Mater 25.18 (2013): 2577-2582.
[6] Butler, David L., et al. "Functional tissue engineering for tendon repair: A multidisciplinary strategy using mesenchymal stem cells, bioscaffolds, and mechanical stimulation." J Ortho Res 26.1 (2008): 1-9.
[7] Staiger, Mark P., et al. "Magnesium and its alloys as orthopedic biomaterials: a review." Biomater 27.9 (2006): 1728-1734.
Click here to see previous highlights.
Disclaimer: Our monthly highlights are sneak peeks of what our R & D department is working on. This does not mean we have what is referenced above ready for manufacturing.